Evaluating patient preferences for thermal ablation versus non-thermal, non-tumescent varicose vein treatments
Objective
To measure patient preferences for attributes associated with non-thermal, non-tumescent (NTNT) and thermal ablation (TA) varicose vein treatments.
- Leverage a choice-based conjoint model to determine which attributes impact a patient’s choice for treatment and the resulting preferred endovenous therapy, assuming equal cost and unrestricted choice
- Determine relative importance of each attribute in the treatment decision
- Understand the trade-offs a patient would be willing to make because of the desired attributes
Patients
(n= 70; three centers, choice-based conjoint, observational)
- Patients currently eligible for treatment or currently being treated were included in the study
- 56% were at the beginning of treatment, 13% mid-treatment, and 31% near the end of treatment
- 70% female; 30% male
- 41% had a household income of <$60,000
Methods
- Each participant received a random version of the survey delivered in person by a study author
- Patient preference was measured for three treatment options: NTNT, TA, or none (to represent compression, weight loss or exercise)
- Active treatment modalities (NTNT and TA) were categorized in the following six attributes:
- Number of injections: 1-3 (NTNT) or 4-7 (TA)
- Number of visits: 3-5 (NTNT) or 4-7 (TA)
- Postoperative pain: barely noticeable (NTNT) or noticeable (TA)
- Risk of an adverse event: 8 in 200 (NTNT) or 1 in 200 (TA)
- Time to return to normal activities: 1-2d (NTNT) or 3-5d (TA)
- Out-of-pocket expenses ($0, $50 or $150)
- Patients were shown ten screens on an iPad that each displayed three hypothetical treatment scenarios for the six attributes – patients did not know the treatment type that was associated
- Patients would then choose one of the available treatment scenarios or none of the above
- Using regression coefficients, investigators estimated willingness-to-pay measures, tradeoff values, and overall value of various attribute combinations based on the treatment selections made
- A market simulation was performed to compare clusters of attributes that mimic NTNT and TA treatments to predict market choice and understand attribute preferences
Key results
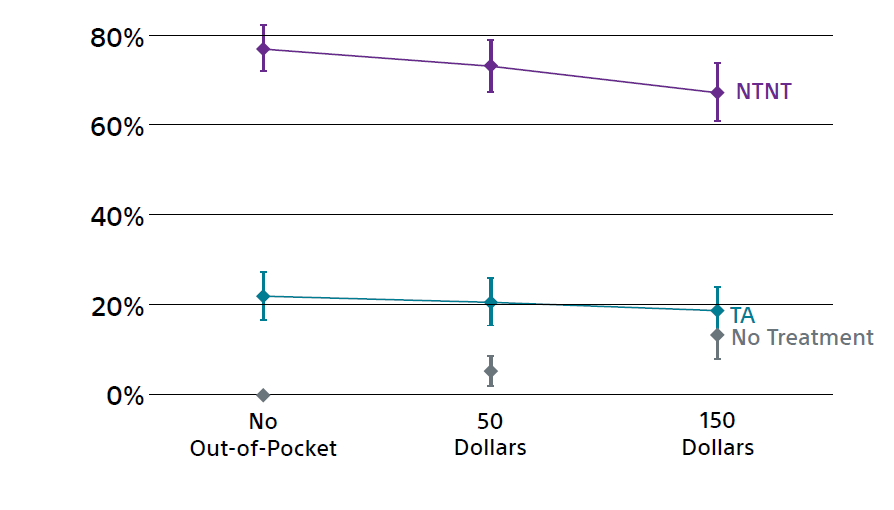
- 60–80% of respondents favored attribute combinations that correspond to NTNT procedures over TA regardless of the level of out of pocket spending
- 14% would forgo treatment with a $150 copay while 6% would forgo treatment with a $50 copay
- The analysis indicates high sensitivity to out of pocket costs for minimally invasive varicose vein treatment and relatively high willingness to pay for reduction in adverse events, postoperative pain, the number of injections, fewer visits and faster return to normal activity
- A few differences were noted by demographics, with males willing to pay more for fewer injections per visit, reduced postoperative discomfort and faster return to normal activity
- Patients with an income of <$60,000 were highly influenced by cost with relative importance of over 50%
Relative importance of each attribute
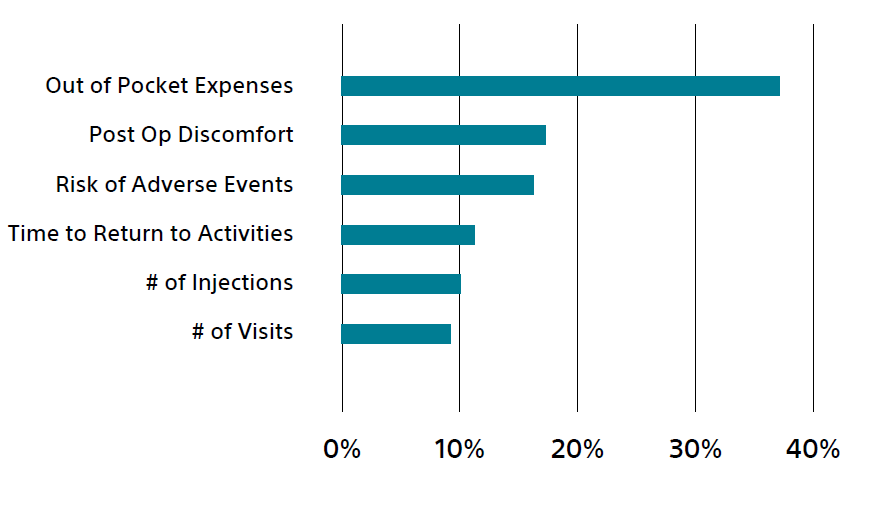
Overall market preference
Limitations
- The study was conducted at three locations in New Jersey; results may be regionally specific
- Scores are sensitive to the levels chosen in the survey design:
- Choosing $100 vs $200 instead of $1 vs $100 could impact results
- A pilot was conducted to inform survey choices
References
1. The study was supported by a grant from Boston Scientific. Boston Scientific had no involvement in the study design; collection, analysis, and interpretation of data; manuscript writing; or the decision to submit the report for publication. Authors, G.D and C.G., are consultants for BTG International.